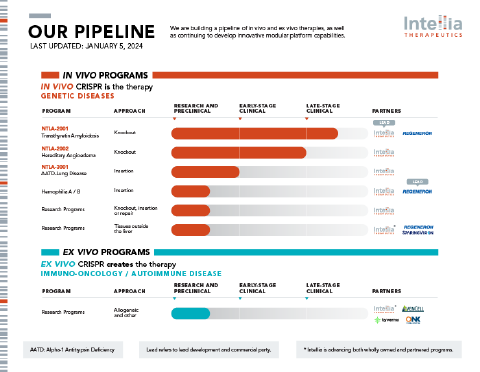
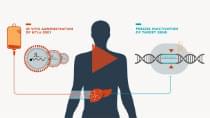
We are building a pipeline of in vivo and ex vivo therapies, as well as continuing to develop innovative modular platform capabilities.
Preclinical
Clinical
Clinical
- AATD: alpha-1 antitrypsin deficiency
- Lead refers to lead development and commercial party
- * Intellia is advancing both wholly-owned and partnered programs.
- ** Hemophilia A program is in the research stage.
- *** This program is being advanced by Regeneron. Intellia is eligible for milestones and royalties.
Download
Learn more