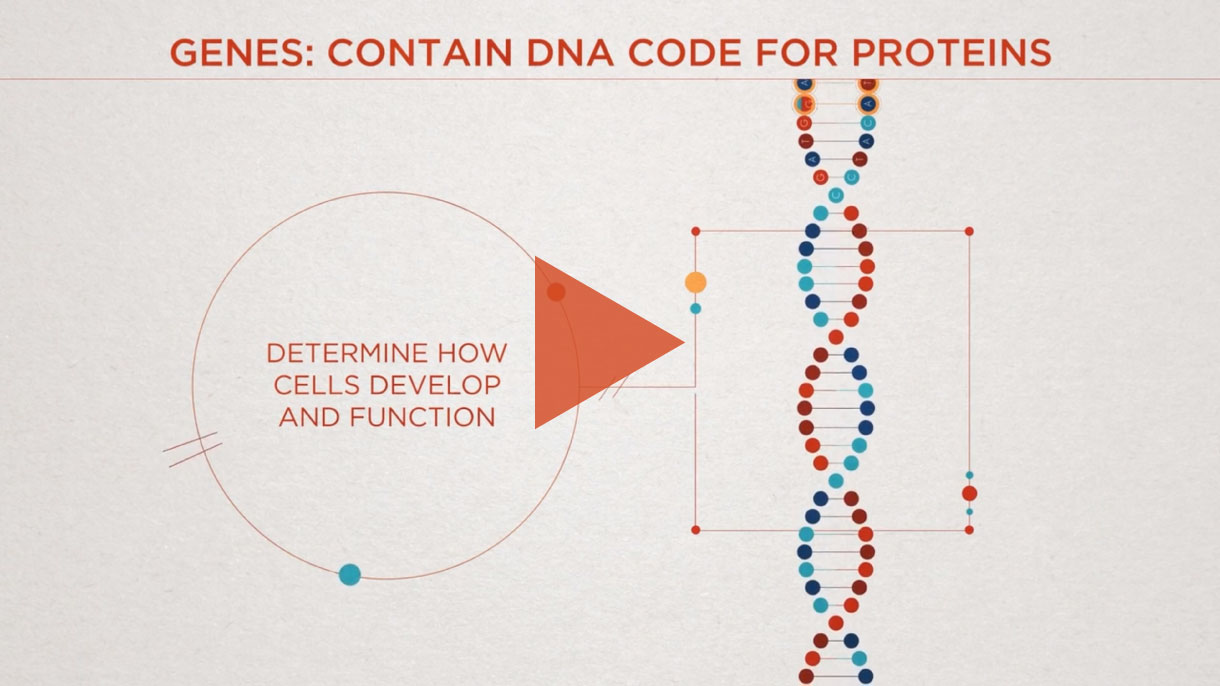
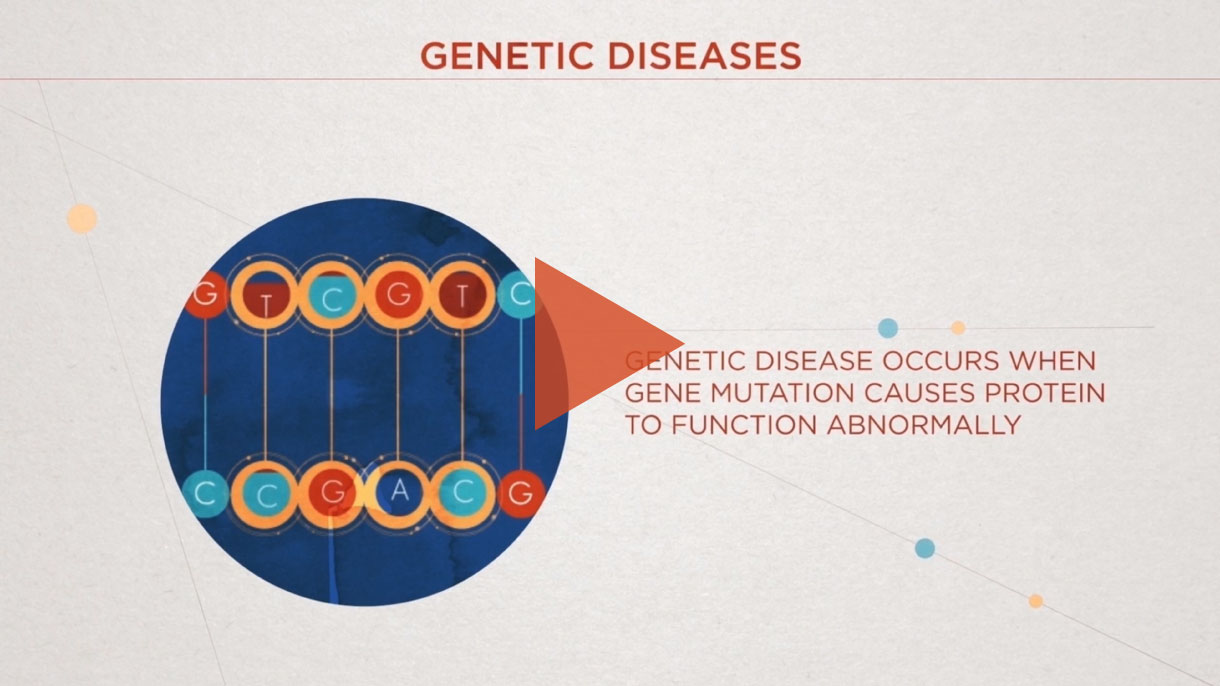
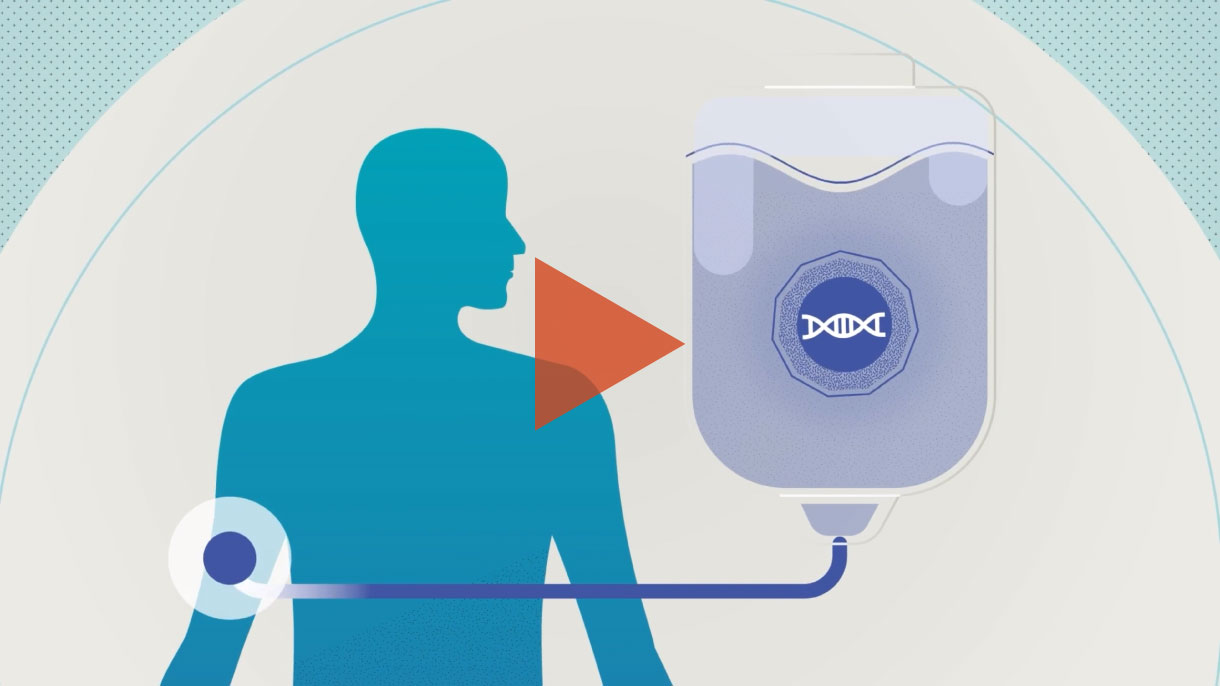
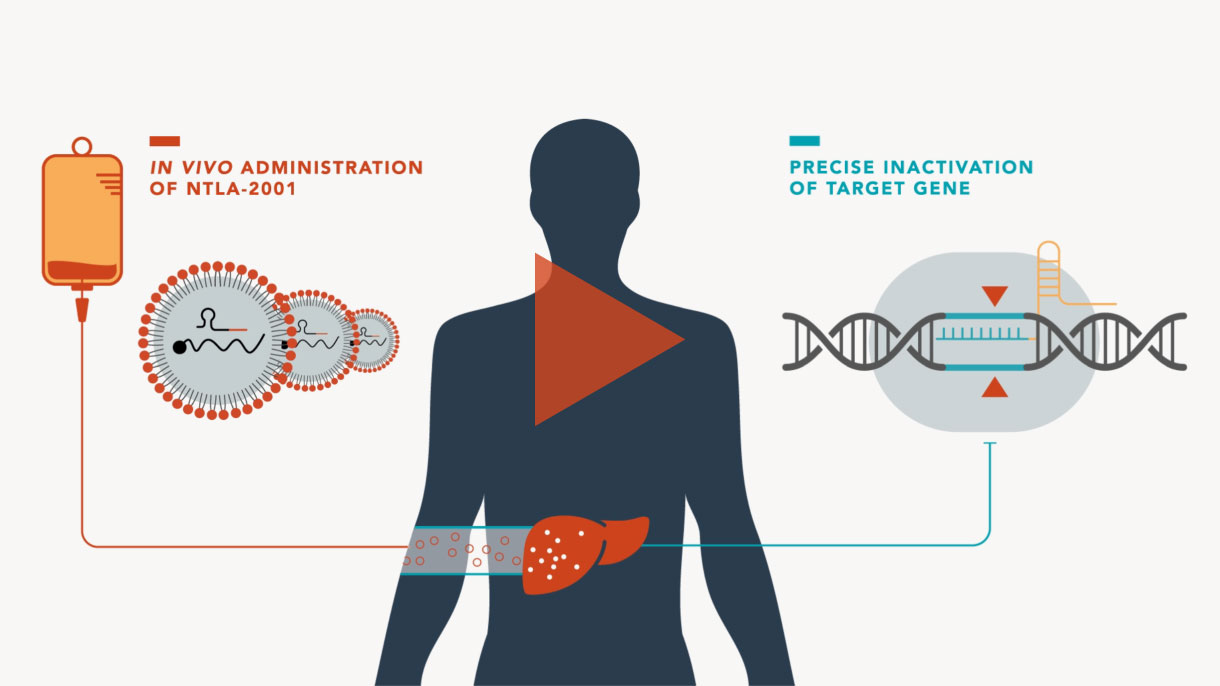
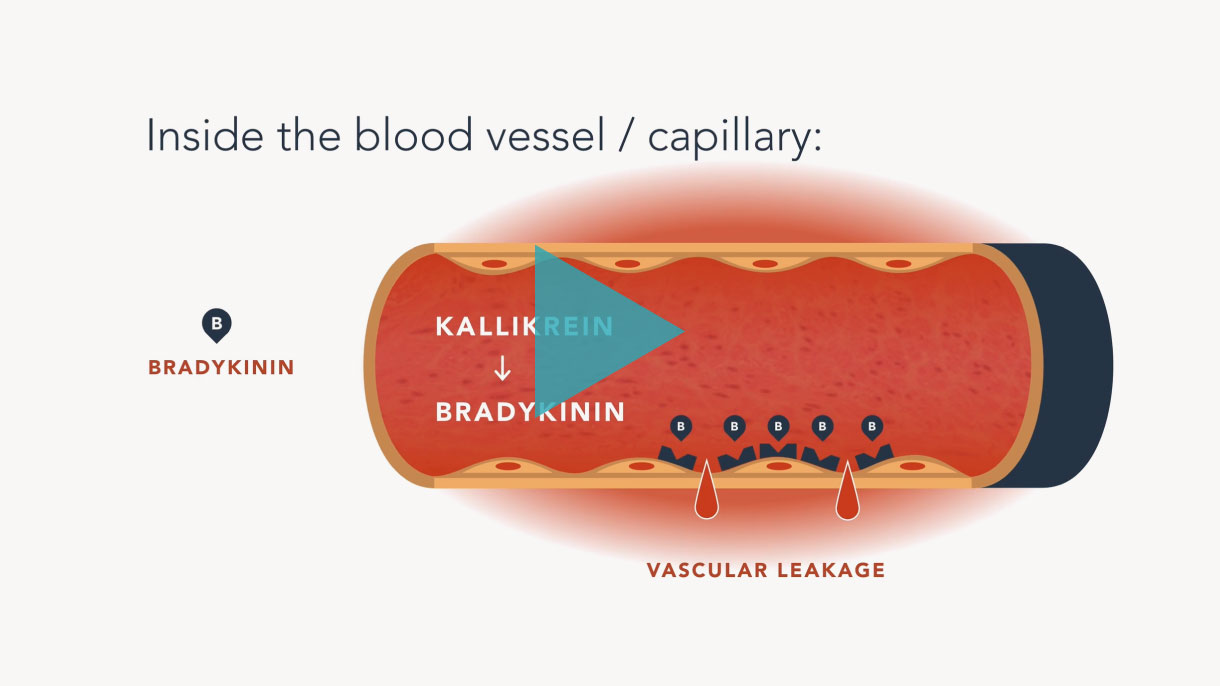
CRISPR/Cas9 is a Nobel Prize-winning gene editing technology that leverages the body’s natural processes to precisely edit DNA.
It has been around for billions of years, initially used by bacteria as part of their immune system to ward off viruses. Today, scientists at Intellia are developing this technology to make targeted changes to genes directly implicated in human disease.
In the future, Intellia hopes to use its CRISPR technology to address a variety of severe, life-threatening diseases.